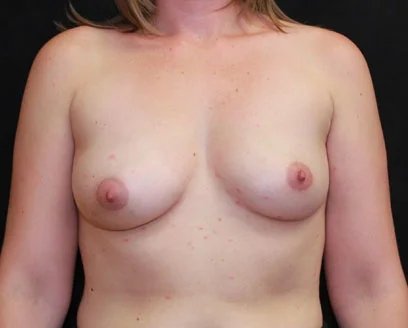
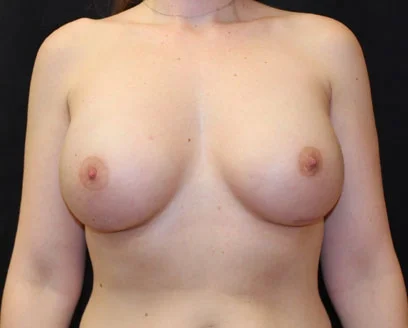
Breast implants (silicone gel or saline – salt water filled) have been used successfully for many years and newer designs have lead to a more natural appearing breast. Recent studies have demonstrated that a woman with silicone implants is no more likely than the rest of the population to develop cancer or immunologic diseases. In other words, there is no scientific evidence that silicone breast implants increase the risk of disease. Satisfied with these studies, in November 2006, the FDA lifted the moratorium on gel implants, allowing their use for both reconstructive and cosmetic breast surgery.
Generally, this is a 2-stage procedure. Typically the 1st stage involves placement of a tissue “expander” under the skin and pectoralis major muscle of the chest wall. Recent trends have also demonstrated successful reconstruction and avoidance of muscle trauma by placing the expander/implant in front of the muscle in select patients. Dr. Parva, board certified plastic surgeon, also uses an AlloDerm graft to reduce trauma to other tissues and to support the expander. The expander procedure can be performed at the time of mastectomy or at a later date as a delayed reconstruction. Over the next 4 to 6 weeks, saline solution is placed into the expander, during office visits, until sufficient expansion of the skin has occurred to create a breast mound. Over the next several months, time allows for the breast skin to soften. If chemotherapy or radiation therapy is indicated, it is often performed during this time. At the 2nd stage, the expander is removed and replaced with the long term breast implant, silicone gel or saline. An alternative approach available to some women is to use a single stage expander/implant that is left in place after being expanded to the appropriate size. This may avoid the second stage surgical procedure necessary when a temporary expander is used. Dr. Parva most often prefers the 2-stage approach and discusses his recommendations with our patients at the initial consultation.
“I was really afraid of having my procedures done but even more frightened of not going through with them & then dealing with the possibility of getting cancer a 3rd time.”
Schedule Your Implant Reconstruction Consultation
Please contact Parva Plastic Surgery for your implant reconstruction consultation with Dr. Parva, board certified plastic surgeon, today.